Search

Researchers are on the hunt for young people with Type 1 Diabetes to participate in a short at-home trial looking at insulin and carbs.

The ENDIA study — Australia’s largest study into the causes of Type 1 Diabetes— reached a significant milestone when it recruited its 700th patient in Perth.

Kids living with Type 1 Diabetes were literally blown away when the Westpac Lifesaver Rescue Helicopter dropped in at the Diabetes WA and PMH kids' camp.

Meet 11-year-old Lucia, who was diagnosed with Type 1 Diabetes when she was 10. Read all about her journey and how she manages her disease in her own words.

Meet Jake O’Brien, the first patient to take part in the longest and largest at-home trial of a hybrid closed-loop insulin pump.

Children's Diabetes Centre co-directors Dr Tim Jones and Dr Liz Davis discuss the importance of the Centre in Impact magazine.
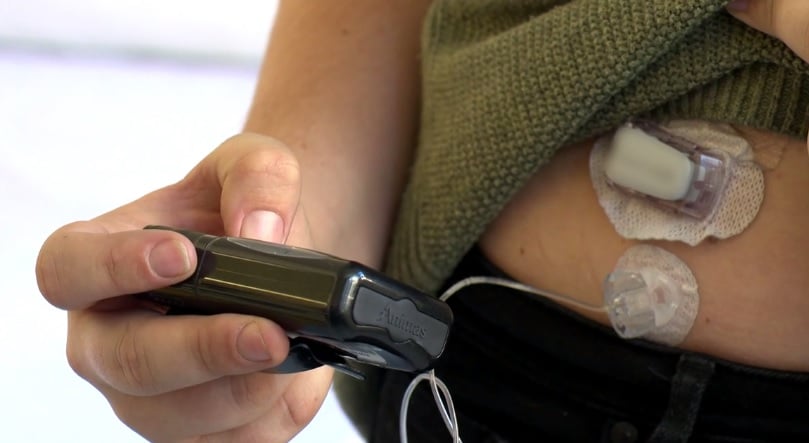
The PMH Diabetes Clinic welcomes the recent Federal Government announcement making fully subsidised Continuous Glucose Monitoring available to young people.

Centre co-director Dr Liz Davis talked to JDRF Australia about women and science to celebrate the recent International Day of Women and Girls in science.

London Olympics torchbearer Gavin Griffiths is living proof that a Type 1 Diabetes diagnosis is no obstacle to making the most out of life.

End-of-year exams may be far from the minds of students managing diabetes but now is the time to get the process started for obtaining special ATAR provisions.
