Search

Ever wondered why your child is grouped into a specific diabetes clinic?Clinical Nurse Consultant Liz Broad said clinics were firstly grouped as metropolitan or regional and then grouped by age.
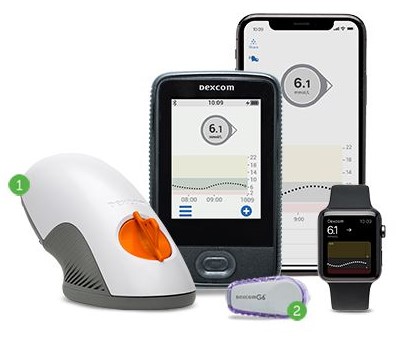
A new Q&A series focusing on the different research themes within the Children’s Diabetes Centre - technology.

Our researchers want to know if starting a gluten-free diet reduces daily glycaemic variability in children and young people with both type 1 diabetes and newly-diagnosed coeliac disease.

This University of Western Australia study, being conducted by a PhD student who is working with the Children's Diabetes Centre, aims to determine for the first time whether head out of water immersion or water-based exercise affects blood glucose levels in people with type 1 diabetes.

February 22, 1999 will forever be etched into the memory of Thomas Johns as it marks the day he became the first child in Australia to be fitted with an insulin pump.

The Kids Discovery Centre is celebrating National Science Week 2020 (August 17-21) with several exciting programs
We sought research experiences of caregivers and their children were enrolled in the Environmental Determinants of Islet Autoimmunity (ENDIA) study.
The National Institute for Clinical Excellence updated guidance for continuous glucose monitoring (CGM) in 2022, recommending that CGM be available to all people living with type 1 diabetes. Manufacturers can trade in the UK with Conformité Européenne (CE) marking without an initial national assessment. The regulatory process for CGM CE marking, in contrast to the Food and Drug Administration (FDA) and Australian Therapeutic Goods Administration (TGA) process, is described.

Our goal is to accelerate the dissemination and implementation of evidence-based models of care for children and young people living with Type 1 Diabetes.

Community Involvement and Consumer Representatives are a really important part of our research. Find out what they have been working on.
